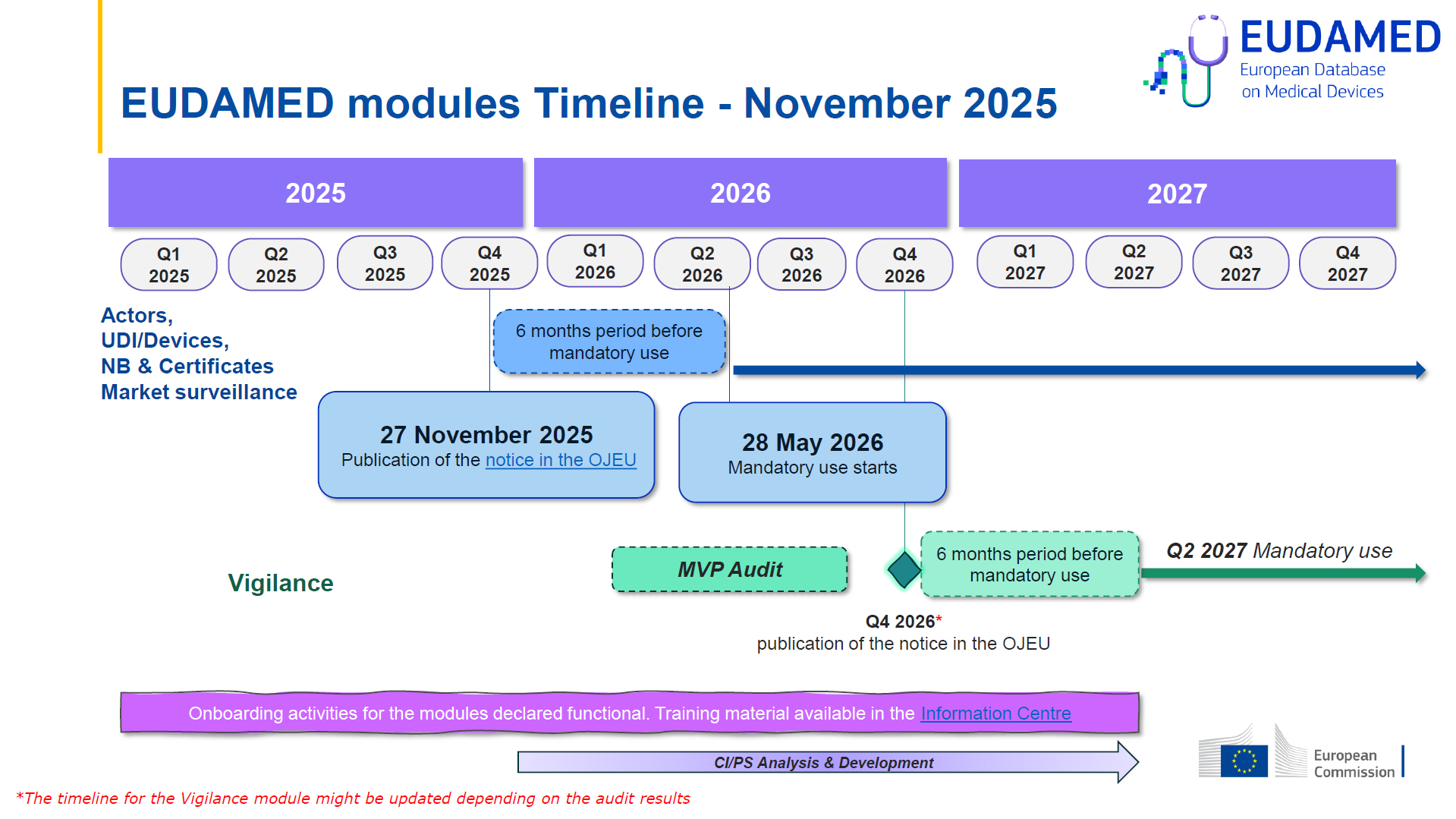
EUDAMED mandatory from 28 May 2026

EUDAMED will finally become mandatory as of 28 May 2026!
The official notice was published by the European Commission in the Official Journal of the European Union on 27 November 2025, initiating a six-month transition period that ends on 28 May 2026.
From this date onwards, the first four EUDAMED modules will become mandatory, marking a decisive step for manufacturers, authorised representatives, importers, and notified bodies operating in the European Union.
WHICH MODULES BECOME MANDATORY
As of 28 May 2026, the following modules are required:
- Actor Registration – registration of economic operators (manufacturers, authorised representatives, importers, etc.)
- UDI/Devices Registration – registration of devices and UDI data
- Notified Bodies & Certificates – registration of notified bodies and certificates
- Market Surveillance – tool supporting market surveillance and coordination between Competent Authorities
KEY DATES TO REMEMBER
| Milestone | Date |
| Publication of OJEU notice | 27 November 2025 |
| EUDAMED mandatory (4 modules) | 28 May 2026 |
| OJEU notice for the Vigilance Module | Q4 2026 |
| Vigilance Module becomes mandatory | Q2 2027 |
WHAT CHANGES AND WHY IT MATTERS
- Registration of economic operators becomes mandatory before placing devices on the market; without this registration, other modules cannot be used.
- Registration of new medical devices/UDI becomes strategic for market placement.
- Notified Bodies must upload new certificates from 28/05/2026, and upload existing certificates by 27/05/2027.
- Competent Authorities across EU Member States will have a harmonised tool for market surveillance and coordination.
The mandatory use of these modules represents a major step towards improving transparency, safety, and traceability of medical devices within the European Union. Early preparation is essential to avoid delays, data submission backlogs, and compliance issues!
HOW ALL4COMPLIANCE CAN SUPPORT YOU
All4Compliance supports manufacturers, authorised representatives, and importers in ensuring that their companies and products are fully compliant with the new EUDAMED deadlines. We can help you plan and manage the entire process – from readiness assessment and registration strategy to device data preparation and submission, as well as regulatory consulting and EUDAMED training.
Our goal is to make the implementation of EUDAMED as simple and efficient as possible, reducing the risk of errors, delays, and compliance-related challenges.
Contact us for a fit-to-need proposal, suited to your company’s needs, your products, and to ensure you meet the new mandatory deadlines!

Address
Biocant Park | Núcleo 04 Lote 2 |
3060-197 Cantanhede
Portugal